Background- The Motivation and Derivation of the Heat Equation:
Heat, or the thermal energy of a system, is generally defined as the solution of The Heat Equation,
is a constant.
It should be noted, however, that this is a special case of a more general PDE which describes the diffusion of some quantity through a medium
is spatially dependent.
For this reason, the Heat Equation is often referred to as The Diffusion Equation. That said, consider the case of a metal sheet made of varying concentrations of different alloys. The Diffusion Equation, as applied to measure heat, cannot be reduced to the general case where is constant. From here, the motivation for the solution to a PDE of these types is clear. It is, however, not clear, how such a description for the physical phenomena of heat flow was derived.
To show the derivation of the Heat Equation let us consider how we might describe the following system:
There is a rod of length , cross-sectional area
, and is uniform in both density
and composition(hence, uniform specific heat constant
and thermal conductivity
), with a heat source attached to one end. Assuming that the rod is perfectly laterally insulated, that is to say, none of the heat is lost to the surrounding environment; and, that the heat source instantaneously heats only one end of the rod. What is the heat of the rod at any given moment and position after the heat source is turned on and instantly back off?
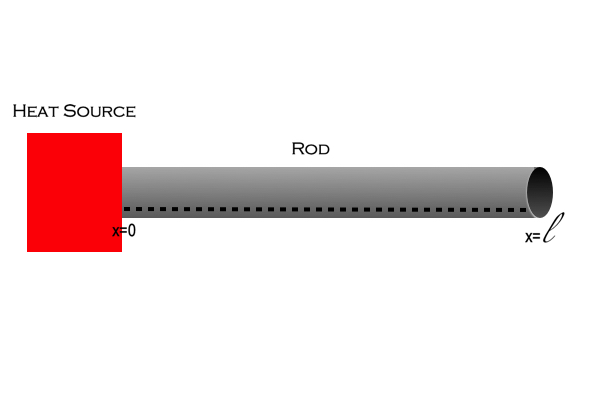
Let’s consider the segment of length ,
, some arbitrary time after the heat source was activated. The total heat within the segment can be written
where is the thermal energy/heat of the rod at some point
and time
. Hence, by the principle of conservation of heat and Fourier’s Law, we find
(1)
where is some external heat source.
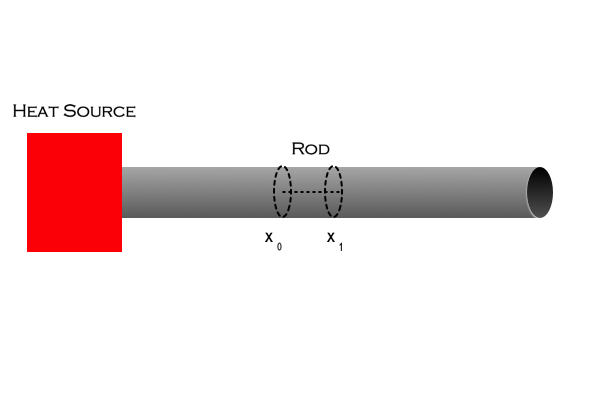
Replacing with
, and applying the Mean Value Theorem we find that equation (1) can be rewritten as,
,
where . After rearranging our terms and simplifying,
Notice, however, we claimed our heat source was instantious. That is, for
by design. Hence, in the limit as
we see that,
(2),
or,
Which, as the rod was laterally insalated, we claim that for the given system equation (2) is equivalent to,
.
Example of the Maximum Principle as a Physical Phenomena:
Consider the case where a rod is connected to a heat source as before. Consider the distribution of heat through the system after some time. We should expect a graph of heat as a function of position at some time , given
, to look something like those in the figure below.
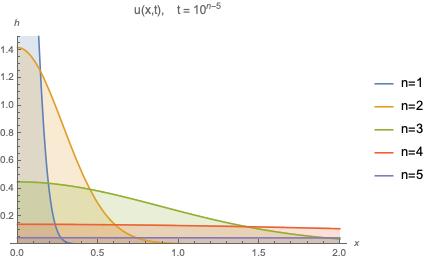
Here we can clearly see that for . This can be interpreted as the physical manifestation of the maximum principle.
Similarly, let’s consider heat at some point over time. We should expect, a graph of heat as a function of time to look something like those in the figure below.
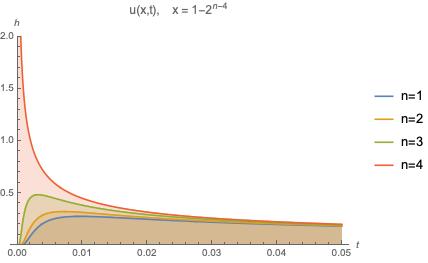
From this we can see that given . This too, can be interpreted as the physical manifestation of the maximum principle.
For a copy of this:
For a copy of the notes used:
For a more “continuous” view of the evolution of heat…
…across the rod:
For a more “continuous” view of the evolution of heat…
…across time:
